Menu
- +Products
- +Allergen testing
- +Culture Media - reagents and reference material
- +Environmental testing solutions
- +Laboratory Consumables
- +Laboratory equipment
- +Microbiology equipment
- +Neogen® Food Safety Solutions
- +Temperature & Humidity Monitoring
- +Services
- SUPPORT
- News
- +About Us
- Contact


Previous
keyboard_arrow_left
Next
keyboard_arrow_right

HARDY DIAGNOSTICS
SterEM™ R2A Agar, USP, Irradiated, Triple Bagged
W390
CAD$60.00
Irradiated R2A Agar is recommended for enumeration of heterotrophic bacteria in water, especially potable water.
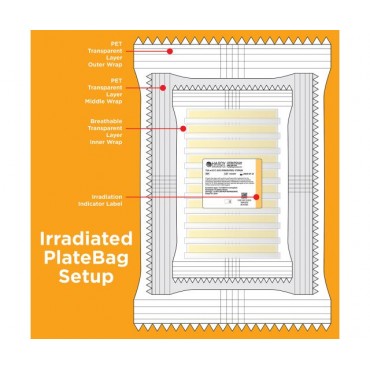
The plates are triple bagged and sterilized by irradiation to promote a higher sterility assurance level.
Features and Benefits:
- Triple bagged - Transparent PET outer and middle bag for distortion-free viewing, breathable transparent inner bag.
- Polystyrene Petri plates feature raised rings for stacking ability and prevent sliding.
- Sterile by irradiation, validated for Sterility Assurance Level (SAL) of 10-6.
- Certificate of Analysis is available on the Hardy Diagnostics website 24/7.
- Manufactured in a licensed cGMP facility with a quality management system certified to ISO 13485.
Download


Previous
keyboard_arrow_left
Next
keyboard_arrow_right
Menu
Filters
Loading...


